The WebApp is sponsored by: 
Extended criteria donors: Opportunity for pediatric liver transplant
Michael Fenlon1, Chinedu Nwudara1, Leonard Aviles1, Austin Forbes1, Michelle Buff1, Motaz Selim1, Michael Zimmerman1.
1Transplant Surgery, University of Utah, Salt Lake City, UT, United States
Introduction:
The survival of children listed for liver transplant (LT) is dependent on the timely availability of a graft with sufficient quality. As the majority of LT are from deceased donor grafts, children are largely in competition with adult candidates. In recent years, rates of adult LT have increased, driven by Extended Criteria Donors (ECD) use. Here, we hypothesize that the increased ECD use adult LT has not been matched in pediatric LT. With the goal of eliminating waitlist mortality, ECD graft use is a potential opportunity to increase the donor pool for pediatric LT candidates.
Methods:
The OPTN STAR file was analyzed, including adult and pediatric deceased donor liver transplants, years 2005-2024. Living donor, multiorgan, and prior transplant patients were excluded. ECD was defined as a donor with: age 50 years or older, macrosteatosis >= 30%, donation after cardiac death, 3+ vasopressors at donation, positive HBV, HCV, HIV serologies, terminal sodium >= 165, total bilirubin >= 4, AST or ALT >= 500. The primary endpoint was patient survival, with a secondary endpoint a composite survival free of: death, graft failure, retransplant, relisting for transplant, persistent bilirubin >= 4.0 within 2 years of LT. Mann-Whitney U, chi-square, Fisher exact tests and Kaplan-Meier survival analysis were performed. A multivariate logistic regression with the Cox proportional hazard model was performed. A p-value <0.05 was considered significant. R 4.5.1 was used for statistical analysis.
Results:
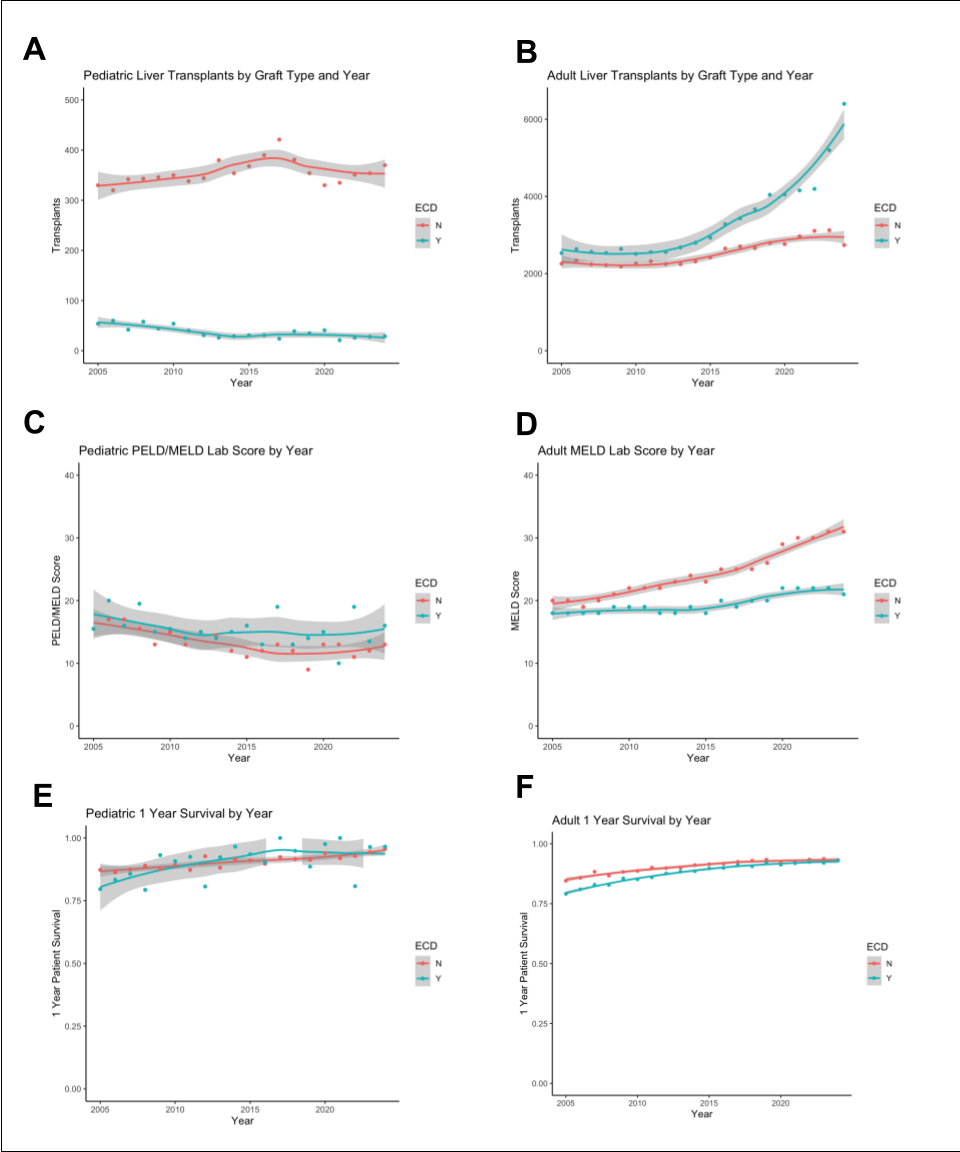
Adults received 50,552 standard (STD) and 67,358 ECD grafts, while children recieved 7099 STD and 743 ECD grafts (125,752 total deceased donor LT). Pediatric ECD use has decreased over time (Figure A) while use in adults has increased (B). Pediatric recipients of ECD grafts were older (5 [1-14], 2 [0-9] years), listed with higher native PELD/MELD lab scores (15 [3-25], 11 [-1-21]), with higher rates of life support at transplant (110/633, 643/6456, n) (medians, [IQR], all p <0.0001). Pediatric 1 year survival has improved, yet remained similar between STD and ECD grafts, while adult ECD outcomes have improved to STD levels (E, F). Multivariate analysis in children showed increasing year transplanted was protective (years 2020-24 0.54-0.87), while life support at transplant (1.69-2.57), recipient age 13-17 (1.39-2.39) and allocation score (1.00-1.02) were risk factors for death (Hazard Ratio 95% confidence intervals, all p< 0.01).
Conclusion:
Given the recently improved outcomes achieved in adult liver transplantation, Extended Criteria Donors should be considered for children.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES