The WebApp is sponsored by: 
Initial Experience with Post-Operative Contrast Enhanced Ultrasound Following Pediatric Whole and Partial Deceased Donor Liver Transplant
Tyler York1, Joshua Badshah1, Antony Attiya1, Toshihiro Nakayama1, Amrita Narang2, Noelle Ebel2, Leina Alrabadi2, Kathryn Czepiel2, Marc L. Melcher1, Thomas A Pham1, Stephan Busque1, Gregory M Tiao1, Erika Rubesova3, Varia A Kirchner1.
1Abdominal Transplantation (Surgery), Stanford University, Stanford, CA, United States; 2Pediatric Gastroenterology, Stanford Children's Health, Stanford, CA, United States; 3Pediatric Radiology, Stanford Children's Health, Stanford, CA, United States
Introduction: Prompt radiographic evaluation is essential for early recognition of vascular complications following pediatric liver transplant (LT). Bedside contrast enhanced ultrasound (CE-US) is a non-invasive modality that avoids radiation exposure, the need for sedation and transport, and has potential to detect early post-LT vascular complications.We describe our initial experiences with its use as a screening and for-cause (FC) modality.
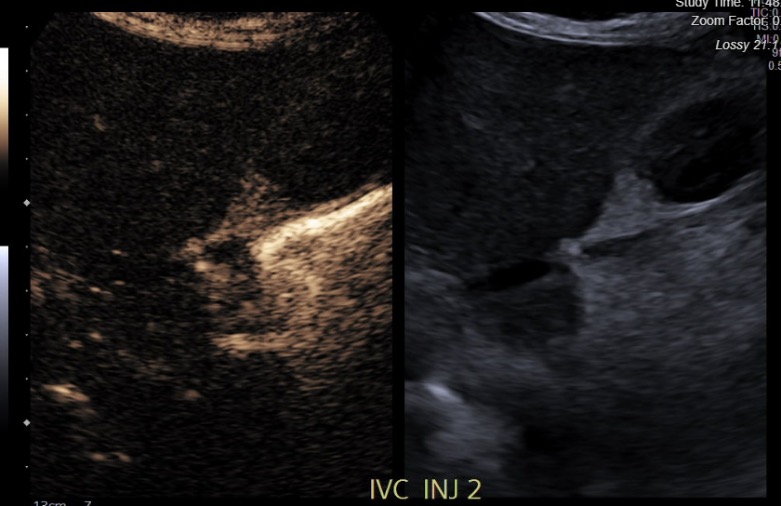
Methods: Retrospective analysis was performed on 20 CE-US in 14 pediatric patients undergoing partial (n=5) or whole graft (n=9) LT, between October 2024 and January 2026 at our institution. Intravenous LUMASON® (sulfur hexafluoride lipid-type A microspheres) was administered per protocol. Demographic, clinical, and radiographic data were analyzed. CE-US findings were correlated with standard of care imaging.
Results: Mean patient age was 7 years old and 57% were females. Indications for transplant included 5 Primary Cholestatic Conditions, 1 Glycogen Storage Disorder, 1 Giant Cell Hepatitis, 2 Autoimmune Hepatitis, 2 Unresectable Hepatic tumors, 2 Acute on Chronic Liver Failure, and 1 Cystic Fibrosis. Compared to standard of care imaging, 6 FC CE-US accurately identified 7 of 8 abnormalities (decreased arterial flow (n=1), asymptomatic arterial anomaly (n=1), IVC stenosis/thrombosis (n=4), parenchymal hypoperfusion (n=1) with 87% sensitivity and 100% specificity. 1 arterial stenosis diagnosed on CTA was not identified on FC CE-US. 4 findings on FC CE-US helped change management. 3 of 14 screening CE-US demonstrated one previously known asymptomatic arterial anomaly and 2 cases of initial poor parenchymal perfusion. The timing of post-operative CE-US varied (mean day post-LT 2.2). There was no standardized reporting of CE-US findings. Contrast enhancement added $90 per study.
Conclusions: Bedside CE-US was safe and affordable with high sensitivity and specificity in pediatric LT recipients. It provided effective and timely diagnosis avoiding the need for anesthesia and transport. Further efforts necessitate prospective study with correlation to standard of care imaging and clinical outcomes, as well as need for the standardized reporting of CE-US findings.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES