The WebApp is sponsored by: 
BOLD and BOLD-EXT, the first global phase III clinical program to evaluate the long-term efficacy and safety of odevixibat in infants with biliary atresia after Kasai hepatoportoenterostomy – study design and current status
Saul J Karpen1, Wendy Sevilla51, Piotr Czubkowski55.
1Stravitz-Sanyal Institute for Liver Disease and Metabolic Health, Virginia Commonwealth University, Richmond, VA, United States; 2Guangzhou Women’s and Children’s Medical Center, Guangzhou, China; 3Pediatric Gastroenterology and Hepatology, Department of Paediatrics, University Medical Center Groningen, University of Groningen, Groningen, Netherlands; 4Hospital for Sick Children, Toronto, ON, Canada; 5Children's Hospital of Fudan University, Shanghai, China; 6Hospital Universitario Vall d'Hebron, Barcelona, Spain; 7Division of Gastroenterology, Boston Children's Hospital, Boston, MA, United States; 8Division of Pediatric Gastroenterology, Hepatology and Nutrition, Stanford University School of Medicine/Lucile Packard Children’s Hospital, Palo Alto, CA, United States; 9Pediatric Gastroenterology and Hepatology, University Children’s Hospital Tübingen, Tübingen, Germany; 10Starship Child Health, Te Toka Tumai, Auckland, New Zealand; 11Centre Hospitalier Universitaire Sainte-Justine, Montreal, QC, Canada; 12Division of Gastroenterology, Hepatology and Nutrition and the Department of Pediatrics, Children's Hospital if Philadelphia and Perelman School of Medicine at University of Pennsylvania, Philadelphia, PA, United States; 13Pediatric Gastroenterology and Hepatology, Hassenfeld Children’s Hospital, NYU Langone, New York, NY, United States; 14Department of Gastroenterology, Royal Children's Hospital, Melbourne, Australia; 15Children's Healthcare of Atlanta, Emory University, Atlanta, GA, United States; 16University of Colorado School of Medicine, Children’s Hospital Colorado, Aurora, CO, United States; 17Hépatologie, Gastroentérologie et Nutrition Pédiatriques, Hôpital Femme Mère Enfant, Hospices Civils de Lyon, Lyon, France; 18The Children’s Hospital at Westmead, Westmead, Australia; 19Semmelweis University, Department of Pediatrics, Budapest, Hungary; 20Hospital Raja Perempuan Zainab II, Kota Bharu, Malaysia; 21Schneider Children’s Medical Center of Israel, Gray Faculty of Medical and Health Sciences, Tel-Aviv University, Tel-Aviv, Israel; 22The Children’s Hospital at Montefiore, Bronx, NY, United States; 23Indiana University/Riley Hospital for Children at IU Health, Indianapolis, IN, United States; 24Department of Pediatrics, Washington University School of Medicine, St. Louis, MO, United States; 25Hépatologie et Transplantation Hépatique Pédiatriques, Centre de Référence de l’Atrésie des Voies Biliaires et des Cholestases Génétiques, Paris, France; 26Department of Paediatric Gastroenterology, Akdeniz University, Antalya, Turkey; 27Department of Paediatrics, University of Malaya, Malaya, Malaysia; 28Mount Sinai Hospital, New York, NY, United States; 29Rady Children's Hospital, San Diego, CA, United States; 30FSMR FILFOIE, ERN RARE LIVER, Hôpital Bicêtre, AP-HP, Université Paris-Saclay, Paris, France; 31Department of Pediatrics Endocrinology, Gastroenterology and Metabolic Medicine, Charité Universitätsmedizin Berlin, Berlin, Germany; 32Paediatric Hepatology, Gastroenterology, and Transplantation, Azienda Ospedaliera Papa Giovanni XXIII, Bergamo, Italy; 33U.O. di Gastroenterologia ed Epatologia Pediatrica, Padua, Italy; 34Pediatric Gastroenterology-Hepatology-Nutrition Unit, Hôpital Universitaire Necker-Enfants Malades, Paris, France; 3534University Lille, Reference Centre for Rare Oesophageal Diseases, Lille, France; 36Division of Pediatric Gastroenterology, Hepatology, and Nutrition, Hacettepe University Faculty of Medicine, Ankara, Turkey; 37Department of Pediatrics, Children's Mercy–Kansas City, Kansas City, MO, United States; 38University of California, San Francisco, CA, United States; 39Primary Children's Hospital, University of Utah, Salt Lake City, UT, United States; 40Nemours Children's Hospital, Delaware, Wilmington, DE, United States; 41Pediatric Gastroenterology and Hepatology, Hannover Medical School, Hannover, Germany; 42Pediatric Gastroenterology Unit, Regina Margherita Children’s Hospital, Azienda Ospedaliera-Città della Salute e della Scienza di Torino, Turin, Italy; 43Birmingham Women’s and Children’s Hospital, Birmingham, United Kingdom; 44Division of Gastroenterology, Hepatology and Nutrition, The Children's Hospital of Pittsburgh, Pittsburgh, PA, United States; 45Oregon Health Science University School of Medicine, Portland, OR, United States; 46Ann & Robert H. Lurie Children's Hospital, Chicago, IL, United States; 47Cincinnati Children's Hospital, Cincinnati, OH, United States; 48Leeds General Infirmary, Leeds, United Kingdom; 49National Taiwan University Hospital, Taipei, Taiwan; 50Childrens Hospital Los Angeles, Los Angeles, CA, United States; 51Ipsen, Cambridge, MA, United States; 52Ipsen, Boulogne-Billancourt, France; 53Albireo, an Ipsen company, Gothenburg, Sweden; 54Ipsen, London, United Kingdom; 55Department of Gastroenterology, Hepatology, and Immunology, Children’s Memorial Health Institute, Warsaw, Poland
BOLD Study.
Saul J. Karpen1, Jiakang Yu2, Henkjan Verkade3, Vicky Lee Ng4, Shan Zheng5, Jesus Quintero6, Andrew Wehrman7, Amrita Narang8, Ekkehard Sturm9, Helen Evans10, Fernando Alvarez11, Kathleen Loomes12, Nadia Ovchinsky13, Winita Hardikar14, Reuven Zev Cohen15, Shikha Sundaram16, Mathias Ruiz17, Susan Siew18, Antal T. Dezsőfi-Gottl19, Wan Mohamad Noor20, Orith Waisbourd-Zinman21, Debra Pan22, Jean P. Molleston23, Janis Stoll24, Emmanuel Gonzales25, Reha Artan26, Way Seah Lee27, Jaime Chu28, Laura Bauman29, Philip Bufler30, Lorenzo D'Antiga31, Mara Cananzi32, Florence Lacaille33, Madeleine Aumar34, Hayriye Hizarcioglu Gulsen35, Ryan Fischer36, Philip Rosenthal37, Kyle Jensen38, Adebowale Adeyemi39, Ulrich Baumann40, Pier Luigi Calvo41, Chayarani Kelgeri42, James E. Squires43, Henry C. Lin44, Alyssa Kriegermeier45, Michael Rogers46, Alizai Naved47, Huey-Ling Chen48, Rohit Kohli49, Wendy Sevilla50, Roshawn Watson50, Fatine Elaraki51, Christelle Baffie50, Anna Wallebeck52, Jennifer Grace52*, Sofia Ganzha53, Anja Vlahovic50, Piotr Czubkowski54
Introduction: Biliary atresia (BA) is a progressive cholangiopathy without approved pharmacologic treatments. A Kasai hepatoportoenterostomy (HPE) aims to restore bile flow, but many patients develop progressive liver injury; ~70% will eventually require liver transplantation.1 By reducing intrahepatic bile acid retention and diverting from the liver, the ileal bile acid transporter inhibitor odevixibat (ODX) could augment and sustain the native liver protective effects of Kasai HPE. BOLD is the first and only phase III clinical program investigating the disease-modifying potential of ODX in BA post-Kasai HPE.
Methods: BOLD (NCT04336722) is a placebo-controlled phase III study evaluating ODX (120 μg/kg/day) over 104 weeks in infants who have undergone Kasai HPE at ≤90 days old. Primary endpoint: time from randomization to first occurrence of liver transplant or death (native liver survival [NLS]); secondary endpoints: time to onset of first sentinel event (sentinel event-free survival), total bilirubin levels, serum bile acid levels, and time to pediatric end-stage liver disease score >15. BOLD-EXT (NCT05426733) is an open-label extension study evaluating long-term efficacy and safety of ODX in patients with BA who completed BOLD; study endpoints are in the Figure.
Results: BOLD initiated in July 2020 across 72 sites in 19 countries (N=254 patients) with an estimated completion in mid-2026. BOLD-EXT initiated in 2022 across 47 sites (target enrollment, N=180 patients). Estimated completion is in 2028.
Conclusion: BOLD/BOLD-EXT will determine whether ODX can achieve disease modification, including a sustained effect on NLS, and evaluate its safety profile in infants and young children with BA post-Kasai HPE.
Reference: 1. Hukkinen M, et al. Best Pract Res Clin Gastroenterol 2022;56–57:101764.
Content from The Liver Meeting®; used with permission from the American Association for the Study of Liver Diseases.
Figure. BOLD-EXT study clinical endpoints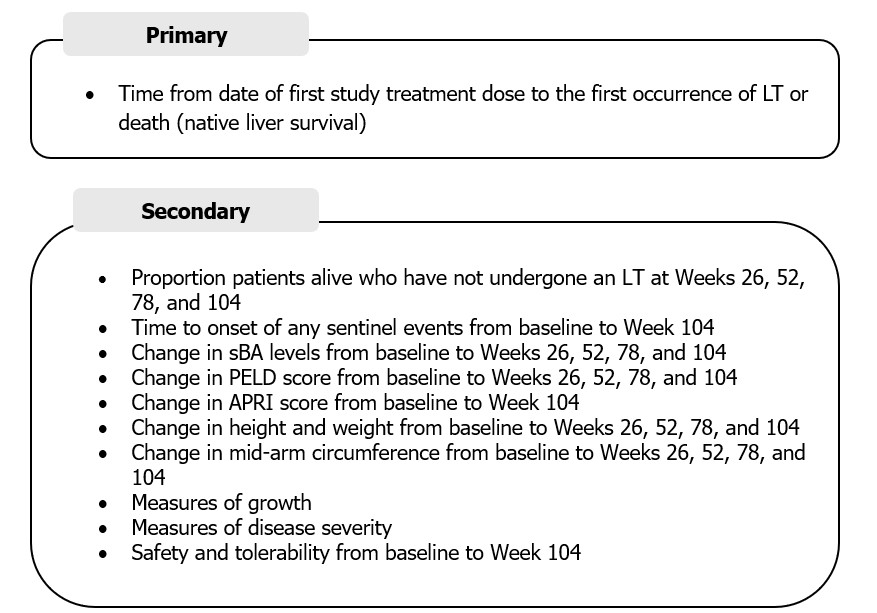
Affiliations: 1Stravitz-Sanyal Institute for Liver Disease and Metabolic Health, Virginia Commonwealth University, Richmond, VA, USA; 2Guangzhou Women’s and Children’s Medical Center, Guangzhou, Ghangdong, China; 3Pediatric Gastroenterology and Hepatology, Department of Paediatrics, University Medical Center Groningen, University of Groningen, the Netherlands;
4Hospital for Sick Children, Toronto, ON, Canada; 5Children's Hospital of Fudan University, Shanghai, China; 6Hospital Universitario Vall d'Hebron, Barcelona, Spain; 7Division of Gastroenterology, Boston Children’s Hospital, Boston, USA; 8Division of Pediatric Gastroenterology, Hepatology and Nutrition at Stanford University School of Medicine/Lucile Packard Children’s Hospital, Palo Alto, CA, USA; 9Pediatric Gastroenterology and Hepatology, University Children’s Hospital Tübingen, Tübingen, Germany; 10Starship Child Health, Te Toka Tumai, Auckland, New Zealand; 11Centre Hospitalier Universitaire Sainte-Justine, Montreal, QB, Canada; 12Division of Gastroenterology, Hepatology and Nutrition, The Children’s Hospital of Philadelphia and Department of Pediatrics, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, PA, USA; 13Pediatric Gastroenterology and Hepatology, Hassenfeld Children’s Hospital, NYU Langone, New York, NY, USA; 14Department of Gastroenterology, Royal Children's Hospital, Melbourne, VIC, Australia; 15Children's Healthcare of Atlanta, Emory University, Atlanta, GA, USA; 16University of Colorado School of Medicine, Children’s Hospital Colorado, Aurora, CO, USA; 17Hépatologie, Gastroentérologie et Nutrition Pédiatriques, Hôpital Femme Mère Enfant, Hospices Civils de Lyon, Lyon, France; 18The Children’s Hospital at Westmead, Westmead, NSW, Australia; 19Semmelweis University, Department of Pediatrics, Budapest, Hungary; 20Hospital Raja Perempuan Zainab II, Kota Bharu, Kelantan, Malaysia; 21Schneider Children’s Medical Center of Israel, Gray Faculty of Medical and Health Sciences, Tel-Aviv University, Tel-Aviv, Israel; 22The Children’s Hospital at Montefiore, Bronx, NY, USA; 23Indiana University/Riley Hospital for Children at IU Health, Indianapolis, IN, USA; 24Department of Pediatrics, Washington University School of Medicine, St. Louis, MO, USA; 25Hépatologie et Transplantation Hépatique Pédiatriques, Centre de Référence de l’Atrésie des Voies Biliaires et des Cholestases Génétiques, FSMR FILFOIE, ERN RARE LIVER, Hôpital Bicêtre, AP-HP, Université Paris-Saclay, Hépatinov, Inserm U 1193, Paris, France; 26Department of Paediatric Gastroenterology, Akdeniz University, Antalya, Turkey; 27Department of Paediatrics, University of Malaya, Malaysia; 28Mount Sinai Hospital, NY, USA; 29Rady Children's Hospital, San Diego, CA, USA; 30Department of Pediatrics Endocrinology, Gastroenterology and Metabolic Medicine, Charité Universitätsmedizin Berlin, Berlin, Germany; 31Paediatric Hepatology, Gastroenterology, and Transplantation, Azienda Ospedaliera Papa Giovanni XXIII, Bergamo, Italy; 32U.O. di Gastroenterologia ed Epatologia Pediatrica, Padua, Italy; 33Pediatric Gastroenterology-Hepatology-Nutrition Unit, Hôpital Universitaire Necker-Enfants Malades, Paris, France; 34University Lille, Reference Centre for Rare Oesophageal Diseases, CHU Lille, U 1286 INFINITE, F59000, Lille, France; 35Division of Pediatric Gastroenterology, Hepatology, and Nutrition, Hacettepe University Faculty of Medicine, Ankara, Turkey; 36Department of Pediatrics, Children's Mercy–Kansas City, Kansas City, MO, USA; 37University of California, San Francisco, CA, USA; 38Primary Children's Hospital, University of Utah, Salt Lake City, UT, USA; 39Nemours Children's Hospital, Delaware, Wilmington, DE, USA; 40Pediatric Gastroenterology and Hepatology, Hannover Medical School, Hannover, Germany; 41Pediatric Gastroenterology Unit, Regina Margherita Children’s Hospital, Azienda Ospedaliera-Città della Salute e della Scienza di Torino, Turin, Italy; 42Birmingham Women’s and Children’s Hospital, Birmingham, UK; 43Division of Gastroenterology, Hepatology and Nutrition, The Children's Hospital of Pittsburgh, Pittsburgh, PA, USA; 44Oregon Health Science University School of Medicine, Portland, OR, USA; 45Ann & Robert H. Lurie Children's Hospital, Chicago, IL, USA; 46Cincinnati Children's Hospital, Cincinnati, OH, USA; 47Leeds General Infirmary, Leeds, UK; 48National Taiwan University Hospital, Taipei, Taiwan; 49Childrens Hospital Los Angeles, Los Angeles, CA, USA; 50Ipsen, Cambridge, MA, USA; 51Ipsen, Boulogne-Billancourt, France; 52Albireo, an Ipsen company, Gothenburg, Sweden; 53Ipsen, London, UK; 54Department of Gastroenterology, Hepatology, and Immunology, Children’s Memorial Health Institute, Warsaw, Poland
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES