The WebApp is sponsored by: 
Terlipressin therapy for portal hyperperfusion secondary to portal vein size discrepancy after pediatric liver transplant
Forum Patel1, Jerilyn Simons1, Kondragunta R Prasad2, Stacee M Lerret1.
1Pediatric Gastroenterology, Hepatology and Nutrition, Medical College of Wisconsin, Milwaukee, WI, United States; 2Transplant Surgery, Medical College of Wisconsin, Milwaukee, WI, United States
Introduction: Type 1 Abernathy malformations are rare congenital portosystemic shunts characterized by absent intrahepatic portal veins that can result in hyperammonemia and encephalopathy requiring liver transplantation. Post-transplant portal hyperperfusion and small-for-size syndrome (SFSS) physiology, defined in adults as graft to recipient weight ratio (GRWR) <1.5%, can occur. Overall, pediatric management strategies for portal hyperperfusion and SFSS are poorly defined.
Method: We report a 5-year-old female with Type I Abernathy malformation who underwent deceased donor whole-graft liver transplantation for persistent hyperammonemia, encephalopathy, neurodevelopmental delay, and growth failure. Despite an acceptable GRWR (1.78%), the post-operative course raised concern for portal hyperperfusion and SFSS.
Results: Intraoperative findings demonstrated marked size discrepancy between the donor portal vein and the patient’s large portosystemic shunt. Postoperatively, the patient developed rapid and severe elevations in aminotransferases, INR, and bilirubin without evidence of vascular thrombosis or primary graft dysfunction. Elevated portal vein velocities raised concern for portal hyperperfusion and SFSS. Octreotide (somatostatin analogue) was initiated with partial biochemical improvement and subsequently transitioned to terlipressin (vasopressin receptor agonist leading to splanchnic vasoconstriction) once available. Following terlipressin initiation, biochemical parameters steadily normalized.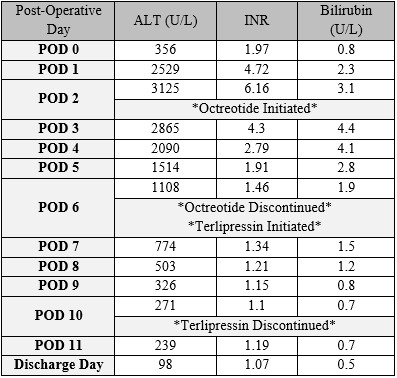 Both therapies were discontinued without recurrence of graft dysfunction. The patient remains clinically well with normal graft function at 18-months post-transplant.
Both therapies were discontinued without recurrence of graft dysfunction. The patient remains clinically well with normal graft function at 18-months post-transplant.
Conclusion: This is the first reported pediatric case of successful use of terlipressin, bridged by octreotide, for management of suspected portal hyperperfusion and SFSS following liver transplantation. Terlipressin may represent an effective noninvasive therapeutic option to modulate portal pressures in select pediatric liver transplant patients.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES