The WebApp is sponsored by: 
Veno-occlusive disease after chimeric antigen receptor T-cell therapy
James Reed1, Stephan A Grupp2, Christine Boone3, Anne Marie Cahill3, Laura S Finn4, Elizabeth B Rand1.
1Gastroenterology, Hepatology and Nutrition, Childeren's Hospital of Philadelphia, Philadelphia, PA, United States; 2Oncology, Children's Hospital of Philadelphia, Philadelphia, PA, United States; 3Interventional Radiology, Children's Hospital of Philadelphia, Philadelphia, PA, United States; 4Pathology, Children's Hospital of Philadelphia, Philadelphia, PA, United States
Veno-occlusive disease (VOD) is seen after stem cell transplant, but is not known as a toxicity of chimeric antigen receptor T-cell therapy (CAR-T). A 7-year-old with 4 prior relapses of B-ALL treated with two remote prior stem cell transplants and two CAR-T treatments was admitted for fever and transferred to ICU 10 days later for rapid progression to acute liver failure (ALF, peak INR 4.25, AST 21,300 U/L, and ALT 5581 U/L) with encephalopathy in the setting of Immune Effector Cell-Associated Hemophagocytic Lymphohistiocytosis-Like Syndrome (IEC-HS) treated with steroids, eculizumab, anakinra, and other agents.
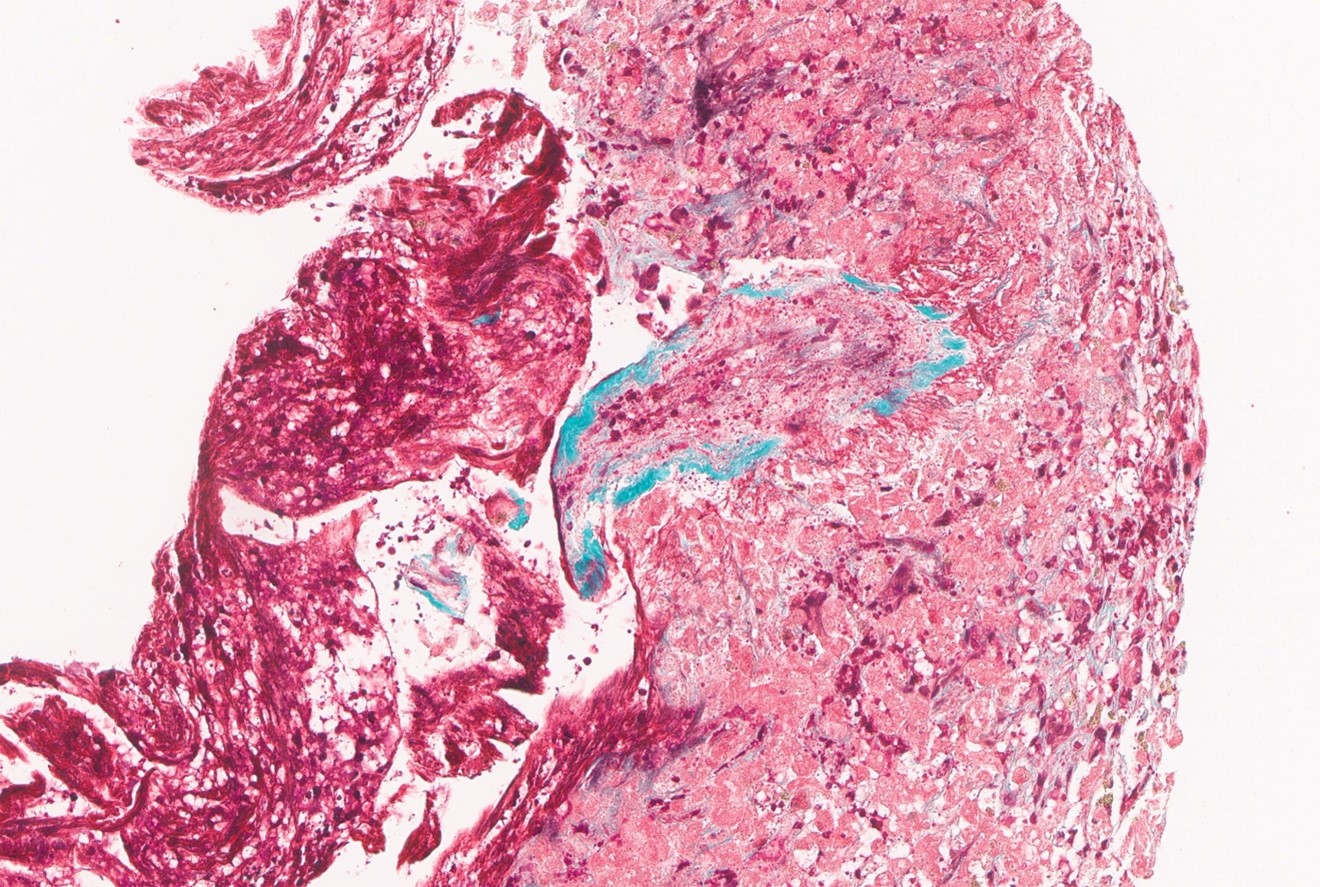
ALF was responsive to ablation of CAR-T cells using cyclophosphamide. She required intubation, vasopressor support, and CRRT. Over 2 weeks, INR declined to <2, allowing decreasing support with blood products. Bone marrow showed B-ALL remission, with presence of some CAR-T cells. She then developed new ascites with PV reversal of flow on US 2 weeks after the onset of ALF. Cholestasis worsened as ascites progressed requiring a peritoneal drain, an overall picture consistent with VOD. Bilirubin peaked at 27.1 almost 2 weeks after ALF declining to 20.9 mg/dL following 10 days of defibrotide. Venogram wedge pressure and endoscopy for variceal bleeding confirmed the presence of portal hypertension. Transjugular liver biopsy demonstrated pericentral necrosis with obstructive changes in hepatic venules but preserved triads, confirming VOD.
She continued to improve and returned to the regular patient unit approximately 6 weeks after presentation. Labs 1 month after conclusion of defibrotide therapy included total bilirubin 8.0 mg/dL and INR 1.55.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES