The WebApp is sponsored by: 
Tacrolimus: Old drug, new tricks to reduce early acute T cell-mediated rejection rates in pediatric liver transplant recipients
Ahmad Salah Sami1, Susan M Stockemer2, Patrick J Healey2, Beatrice E Rosete2, Niviann M Blondet1, Andre AS Dick2, Ryutaro Hirose2, Evelyn K Hsu1, Katelyn Saarela1, Danielle R Wendel1, Sarah E Aker2, Pamela L Valentino1.
1Division of GI and Hepatology, Seattle Children's Hospital, Seattle, WA, United States; 2Division of Transplantation, Seattle Children’s Hospital, Seattle, WA, United States
Introduction:
Acute T-cell-mediated rejection (TCR) remains a significant complication following pediatric liver transplantation (LT). Subtherapeutic tacrolimus exposure early post-transplant is a modifiable risk factor for TCR. This quality improvement (QI) study aimed to reduce early TCR by implementing interventions to achieve therapeutic tacrolimus trough levels (10–12 ng/mL) in the first week post-transplant.
Methods:
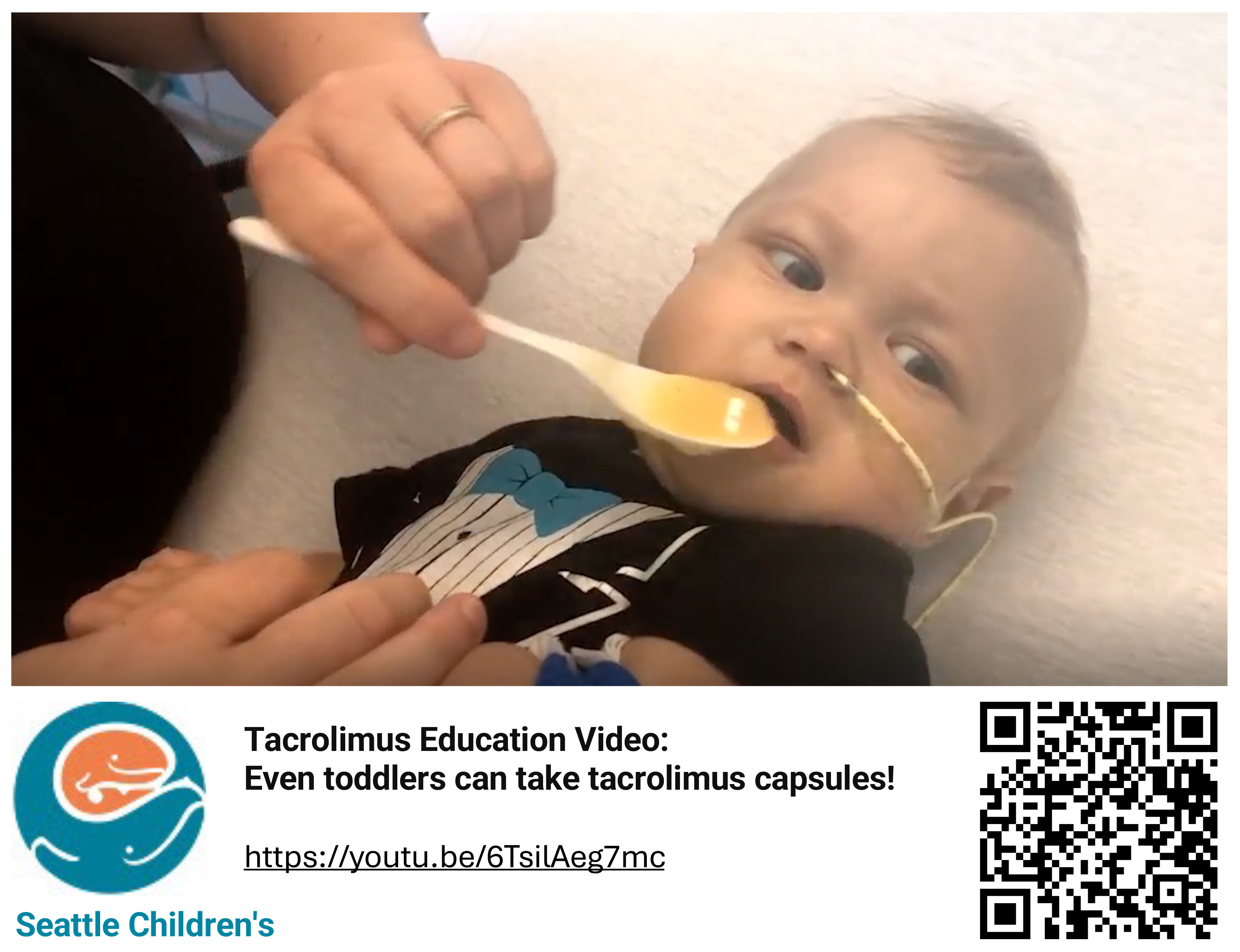
Prospective implementation of standardized approaches to tacrolimus administration between 10/2022-12/2023 (PDSA-Cycle#1) and 1/2024-9/2025 (PDSA-Cycle#2) included: favoring capsule formulation, dose escalation by 20–50% for subtherapeutic levels, structured caregiver and nursing education (including a discharge video, Figure.1), and increased minimum starting doses in PDSA-Cycle#2 (1mg BID) after review of outcomes from PDSA-Cycle#1 (0.5mg BID). Outcome goals included: achieving ≥2 tacrolimus trough levels ≥10 ng/mL between post-operative days (POD) 5-7 in >33% of LT recipients and reducing 90-day TCR rates to <35%. A retrospective analysis of LT recipients from 02/2021-09/2022 informed the baseline performance. Retransplant and combined organ transplant recipients were excluded from the analyses.
Results:
Before intervention, 30% of 27 LT recipients achieved target tacrolimus levels by POD 5-7, with a 90-day TCR rate of 48%. Following QI implementation, 38% of 29 LT recipients achieved tacrolimus targets by POD 5-7 (27% in PDSA-Cycle#1; 50% in PDSA-Cycle#2). 90-day TCR rates declined to 14% overall (22% in PDSA-Cycle#1; 0% in PDSA-Cycle#2).
Conclusion:
A standardized approach to tacrolimus administration and dosing significantly improved early therapeutic exposure and reduced early TCR. Iterative QI adjustments led to progressive outcome improvement, underscoring the importance of optimizing tacrolimus bioavailability in pediatric LT recipients.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES