The WebApp is sponsored by: 
Role of Biologic Therapies in Chronic Allograft Enteropathy in Pediatric Intestinal/Multivisceral Transplantation: A Single Center Experience
Amala Alenchery1, Vikram Raghu MD, MS1, Ahmad Elsabbagh MD2, Ajai Khanna MD2, Armando ganoza MD2, Geoffrey Bond MD2, Jeffrey Rudolph MD1, Jennifer Picarsic MD3, Kyle Soltys MD2, Rakesh Sindhi MD2, George Mazariegos MD2, Simon Horslen MD1, James Squires MD, MS1.
1Division of Gastroenterology, Hepatology and Nutrition, UPMC Children's Hospital of Pittsburgh, Pittsburgh, PA, United States; 2Department of Surgery, Division of Transplantation, University of Pittsburgh, Pittsburgh, PA, United States; 3Professor of Pathology, University of Pittsburgh School of Medicine, Pittsburgh, PA, United States
INTRODUCTION: Chronic allograft enteropathy (CAE) remains a major cause of late graft dysfunction and failure following intestinal/multivisceral transplantation (I/MVT). Management is often challenging with suboptimal outcomes. Biologic therapies targeting immune-mediated inflammation have emerged as potential adjunctive treatments; however, data supporting use is limited.
METHODS: We conducted a retrospective case series of I/MVT recipients diagnosed with CAE and treated with biologics at our institute. Clinical characteristics, indications, pre- and post-therapy histopathology, and treatment regimens were reviewed. Patient and graft survival as well as adverse events were reviewed.
RESULTS: 10 intestinal transplant (isolated bowel-2, liver/intestine-2, multivisceral including liver-6) recipients with CAE who received biologics were identified. 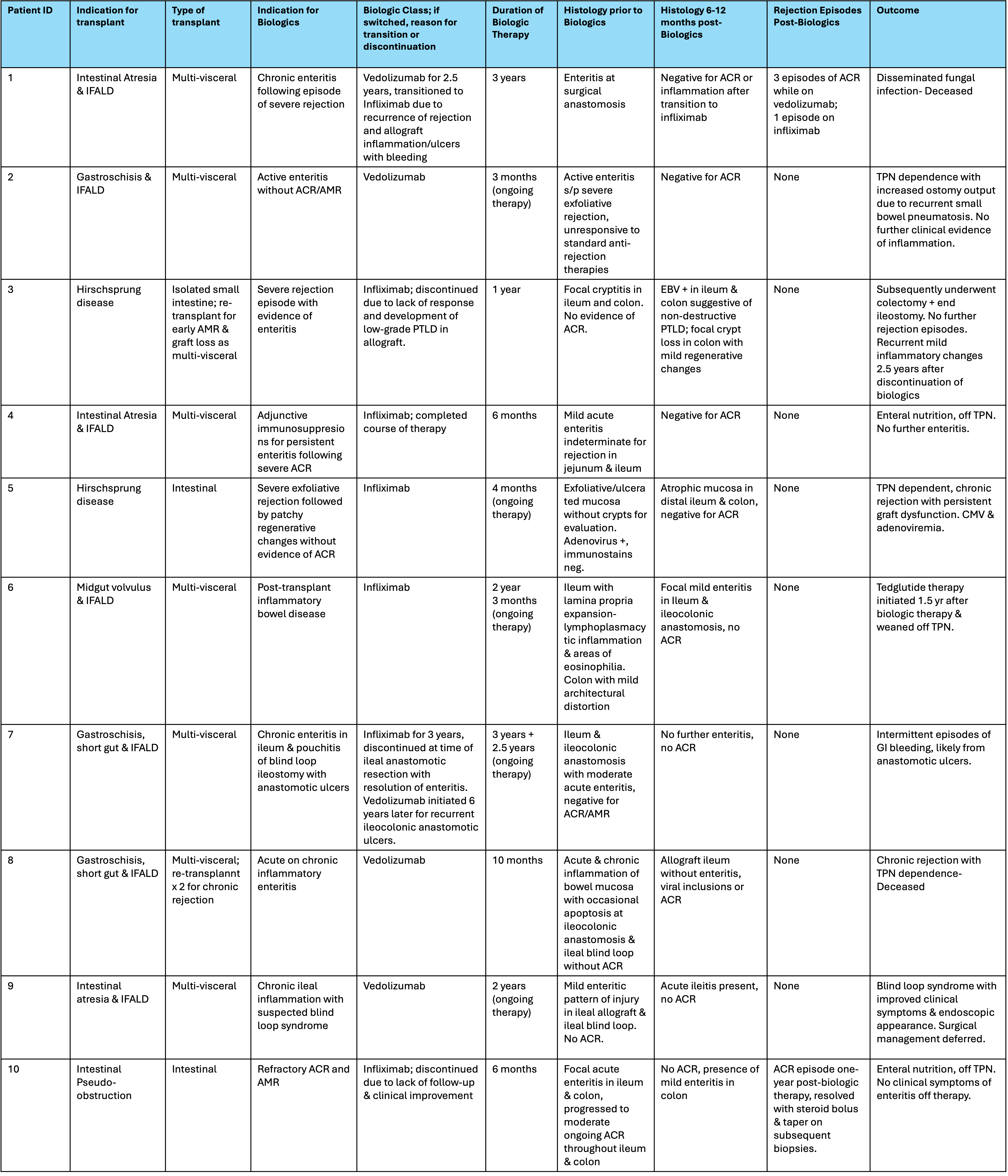 All patients had at least one rejection episode prior to initiation of biologic agents: 6 received anti-tumor necrosis factor(infliximab) and 4 received anti-integrin(vedolizumab). Of the 2 re-transplanted patients, one had chronic rejection needing three MVT and subsequently died. The other had early graft loss from antibody mediated rejection of intestinal transplant with subsequent MVT. Both patients received biologics only after their last re-transplant. 8/10 patients had improved symptoms and stabilization of graft function.One patient developed rejection on biologics. 4/10 showed partial histologic improvement, with disease stabilization in the remainder. Therapeutic classes were switched in 2 patients. There were no unexpected adverse effects.
All patients had at least one rejection episode prior to initiation of biologic agents: 6 received anti-tumor necrosis factor(infliximab) and 4 received anti-integrin(vedolizumab). Of the 2 re-transplanted patients, one had chronic rejection needing three MVT and subsequently died. The other had early graft loss from antibody mediated rejection of intestinal transplant with subsequent MVT. Both patients received biologics only after their last re-transplant. 8/10 patients had improved symptoms and stabilization of graft function.One patient developed rejection on biologics. 4/10 showed partial histologic improvement, with disease stabilization in the remainder. Therapeutic classes were switched in 2 patients. There were no unexpected adverse effects.
CONCLUSION: Biologics were associated with clinical and histologic improvement or stabilization in select I/MVT recipients with CAE, suggesting a potential role as adjunctive therapy. Prospective studies are needed to better define efficacy, optimal patient selection, and long-term safety in this population.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES