The WebApp is sponsored by: 
Cost-Effectiveness of Adolescent-Initiated Semaglutide for Metabolic Dysfunction-Associated Steatohepatitis (MASH): Modeled Implications for Long-Term Liver Transplantation Risk
Tierra L Mosher1,2, Himaja Nagireddy1, Conor Messer1,3, Leandra Bitterfeld1,4, Marwa Abu El Haija2, Rachel Herdes2, Amrita Narang2, Kathryn Czepiel2, Steven M Asch1, Douglas K Owens1, Marissa B Reitsma1.
1Department of Health Policy, Stanford University, Palo Alto, CA, United States; 2Department of Pediatrics, Division of Gastroenterology, Hepatology, & Nutrition, Stanford University School of Medicine, Palo Alto, CA, United States; 3Department of Biomedical Data Science, Stanford University , Palo Alto, CA, United States; 4Intermountain Health, Salt Lake City, UT, United States
Purpose: Semaglutide is approved for adults with metabolic dysfunction–associated steatohepatitis (MASH) and fibrosis, but no pharmacologic treatments are approved for adolescents despite rising prevalence and risk of disease progression. We evaluated the cost-effectiveness of semaglutide initiation in adolescents with MASH and fibrosis versus lifestyle modification.
Methods: We developed a deterministic Markov cohort model simulating lifetime disease progression in U.S. adolescents with MASH. Health states include fibrosis stages F0–F4, decompensated cirrhosis, hepatocellular carcinoma, liver transplantation, and death. Transition probabilities came from Le et al. (2023); treatment effects came from ESSENCE trial data. Costs and utilities came from the literature. From the healthcare sector perspective, we compared lifestyle modification with 72-week semaglutide initiated at age 12 versus 18. Outcomes included lifetime costs (2025 USD) and quality-adjusted life-years (QALYs) over a lifetime horizon with 3% annual discounting, and incremental cost-effectiveness ratios (ICERs) using a $50,000/QALY threshold.
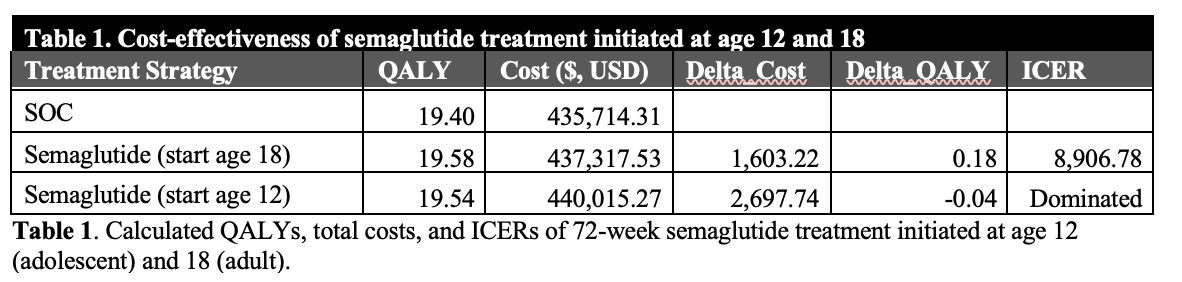
Results: Compared with lifestyle modification, semaglutide reduced progression to advanced fibrosis/cirrhosis and decreased liver transplantations by 3.2%. While adolescent initiation improved outcomes versus lifestyle modification, initiation at age 18 was preferred (ICER $8,906/QALY; Table 1). In sensitivity analyses assuming sustained effectiveness beyond trial follow-up, adolescent initiation became preferred. Treatment remained cost-effective with modest decreases in efficacy and increases in drug costs and became cost-saving at an annual drug price of $2,940.
Conclusion: Short-term semaglutide for adolescents with MASH provides substantial health benefits, with drug costs largely offset by reduced medical costs, supporting adolescent pharmacologic treatment while highlighting the need for more adolescent trial data.
Email: info@splitmeeting.org
If you have any questions during the meeting, please go to the registration desk. Our emails will be monitored sporadically.
REGISTRATION DESK OPENING TIMES